Leave a Reply
Your email address cannot be published. Required fields are marked*
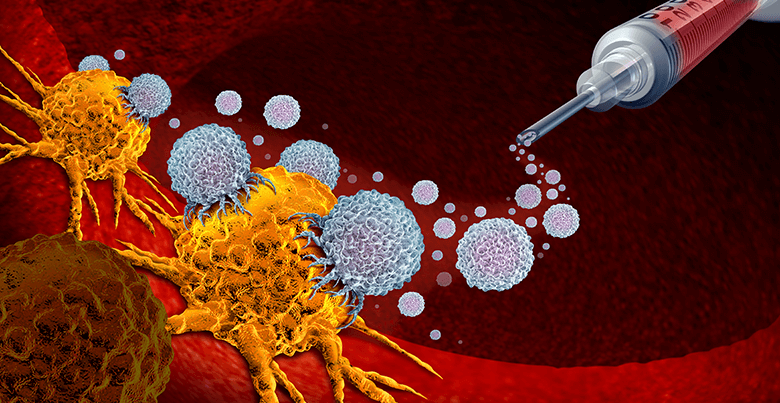
What is Cancer?
In brief, cancer is a condition caused by mutated cells that grow uncontrollably in the body1. These cells are faulty as they did not have the ability to stop dividing and die for cell turnover (apoptosis). There are two types of cancer cells which are benign and malignant1. Benign cells usually cause no harm to the patient and could be removed based on physician’s advice. However, malignant ones are very worrying as the uncontrollable spread of these cells might cause damages to other parts of the patient’s body. Malignant cells also have the ability to hide from the body’s immune system so it can ‘mask’ themselves and hardly be destroyed. There are many known treatments for cancer such as chemotherapy, radiotherapy, surgery and also immunotherapy, which has been actively studied to improve the current treatment.
History of Immunotherapy
Father of Immunotherapy, William B. Coley, who was also an orthopedic surgeon, had attempted to harness the power of the immune system for treating cancer in late 19th century by injecting his patients with “Coley’s toxin”, a mixture of live and inactivated bacteria. This was done in hope to induce sepsis, strengthen the body’s immune system and activated the antitumor responses. Through this, it has become the first ever documented active cancer immunotherapy intervention in history3.
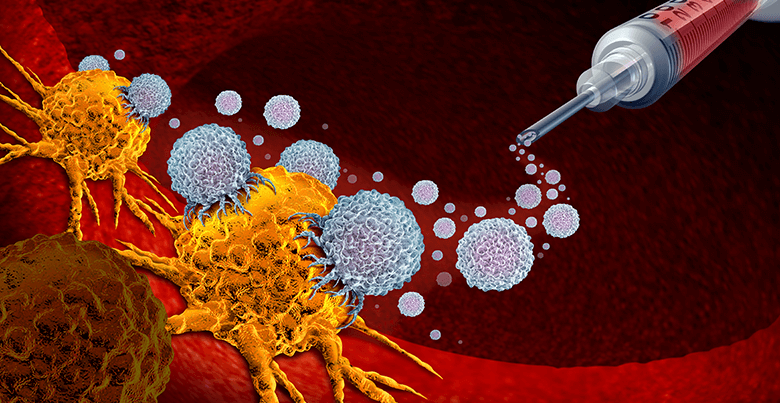
How does immunotherapy help4?
Immunotherapy may educate the immune system to recognize and attack cancer cells without harming the body cells. It can also help to boost immune cells and strengthen the immunity army to attack cancer cells. Besides that, immunotherapy can also be used to provide the patient’s body with additional components (such as antibodies or antigens) to enhance immune response.
Immunotherapy vs Conventional therapy (Chemotherapy)
Chemotherapy is a conventional treatment in treating cancer where it destroys rapidly dividing cells, not specifically targeting cancerous cells but also other cells like hair follicles and gut lining5. These will cause severe side effects to the cancer patient. On the other hand, immunotherapy will only give side effects if there are overstimulation and mistargeted immune response. However, in worst case scenario, immunotherapy may be life-threatening and cause death if the side effects are very severe. So, there are pros and cons in opting for cancer treatment which makes it very important to get the physician’s consultation before deciding which treatment is good for your body as there are many factors to look onto such as, cancer stages, body’s condition, immune system which could be affected by the body’s gut microbiota composition. Gut also represents almost 80% of our entire immune system6. This is due to the fact that these gut microbiomes act as a gatekeeper where it teaches immune cells (T-cells) to distinguish foreign entities from our own tissue7.
Gut microbiota role in our body
Gut microbes, a special “second organ” has a very important role in our body, and a lot of studies have reported that they are usually associated with chronic diseases8. Gut microbiota composition is actually dependent on our lifestyle, nutrition, diet, heredity, and environment. Studies have shown that a series of changes in the gut following the antibiotic treatment can also increase the number of bad bacteria, trigger infections and increase the tumor recurrence9,10,11. Thus, it is good to have a balanced gut microbiota in maintaining good health. The imbalance of gut microbiota can be replenished by consuming probiotics.
Did You Know?
A quarter of 1000 drugs that are commonly used by people may affect the gut microbiota, and 78% of more than 100 antibiotics affect the growth of at least one gut microbe12.
Potential of probiotics/gut microbiota in aiding immunotherapy
In this context, we would like to relate with the benefits of immunobiotics. Immunobiotics are probiotics that have beneficial effects and have the ability to interact with the immune system in our body13. The probiotics in HEXBIO® are also known as immunobiotics based on clinical study tested, which consists of MCP® BCMC® strains (Lactobacillus acidophilus BCMC® 12130, Lactobacillus casei subsp BCMC® 12313, Lactobacillus lactis BCMC® 12451, Bifidobacterium longum BCMC® 02120, Bifidobacterium bifidum BCMC® 02290, and Bifidobacterium infantis BCMC® 02129)14,15. Although some researches have reported that Bifidobacterium and Lactobacillus genera could limit the antitumoral response of Cytotoxic T lymphocytes (a type of immunotherapy)16,17, there are also studies that reported on Bifidobacterium species, which are B. longum and B. breve to have the ability in improving the immunotherapy’s results18,19,20. Besides that, bacteria from the Lactobacillus genus are also responsible in keeping a tolerogenic state of the mucosa and increase the activity of nerve cells (dendritic), both of which support the immunotherapy21,22,23,24,25,26,27,28,29,30. This mucosal immune system (mucosa) in our body has been proven to be improved with the help of the MCP® BCMC® strains, where it stabilizes the mucosal immune function and protect from increased intestinal permeability31. In a way, it has the potential to strengthen the immune system in our body.
Another interesting study conducted by the researchers at the National Cancer Institute (NCI) for Cancer Research, part of the National Institutes of Health (NIH), where they successfully improve the melanoma patients’ immune system to respond to immunotherapy treatment through fecal microbiota transplant32. The study was the first one that has ever been conducted using fecal transplant method. Based on the authors, they took fecal from patients that show positive response to immunotherapy and transplant it into patients that do not have any response to the immunotherapy treatment – to improve the gut microbiota composition in their body that may influence the immune system32. So, from all of these findings, we can say that the current knowledge on this matter is still debatable and may be contradicting with each other, thus, it is highly advisable that cancer patients that are using immunotherapy treatment to get their physician’s insights before deciding to consume probiotics. Also, in looking into this matter, we have done our own study on colorectal cancer patients to evaluate the effectiveness of our own product as different strain have different effects on the human body.
HEXBIO® uses in cancer study
HEXBIO® containing MCP® BCMC® strains have been studied in colorectal cancer (CRC) patients, focusing on their surgical recovery, return of normal gut function, and also those who had gut disturbances after chemotherapy sessions14,15,33. All of the studies were done using a prospective study that measure the effectiveness of a new treatment. These CRC patients were given with HEXBIO® before their scheduled surgery and their conditions were evaluated after the surgery. It was reported that there is 31% faster return of gut function and 50% earlier hospital discharge compared to normal conditions33. Besides that, it was also reported that the use of HEXBIO® on CRC patients help to reduce the inflammatory cytokines in their body, thus, reducing inflammation, reducing side effect after chemotherapy, and significantly improve the patients’ immune system14,15.
How HEXBIO® may be helpful with the side effects of immunotherapy
As mentioned in earlier paragraph, immunotherapy may cause side effects. The most common and less serious symptoms include fatigue (tiredness), diarrhea, fever, shortness of breath, rash and/or blisters, nausea, vomiting, itching, headache, weight loss, difficulty falling or staying asleep (insomnia), and decreased appetite. Other than that, in some cases, it might also cause severe side effects that could be life-threatening. The most reported conditions are inflammation of lung (pneumonitis), liver (hepatitis), colon (colitis), brain (neuropathy, meningitis, encephalitis), kidney problem and/or failure and hormonal gland problems (thyroid, pituitary, adrenal glands and pancreas). Most of these side effects are caused by inflammation in the body that could affect the functionality of the organs. Based on our clinical studies in colorectal cancer patients, it has been shown that HEXBIO® consumption could significantly lower the inflammatory cytokines that are usually related with cancer conditions14,15. When the inflammatory cytokines are reduced, there will be less likely for inflammation to happen and this will prevent serious inflammation in the body. Thus, improve the recovery time of the patient. This could also be related with the function of HEXBIO® in easing gut disturbances to encounter mild symptoms experienced by these patients (vomiting, nausea, diarrhea, insomnia, fatigue etc.) that has been clinically proven to be effective14,15,33. Other than that, HEXBIO® has been tested to improve kidney function and reduce serum urea levels which could help improve renal profile for those with kidney problems among the cancer patients34.
References
[1] National Cancer Institute. Understanding Cancer (Internet). National Institutes of Health (NIH), 2021. Available online: https://www.cancer.gov/about-cancer/understanding/what-is-cancer. Accessed on June 1, 2022.
[2] Cohen E. What is Cancer Immunotherapy? (Internet). Cancer Research Institute, 2020. Available online: https://www.cancerresearch.org/en-us/immunotherapy/what-is-immunotherapy. Accessed on June 1, 2022.
[3] Esfahani K, Roudaia L, Buhlaiga N, Del Rincon SV, Papneja N, Miller WH Jr. A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol. 2020 Apr;27(Suppl 2):S87-S97. doi: 10.3747/co.27.5223. Epub 2020 Apr 1. PMID: 32368178; PMCID: PMC7194005.
[4] American Cancer Society. How Immunotherapy is Used to Treat Cancer (Internet). American Cancer Society Inc., c2022. Available online: https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/what-is-immunotherapy. Accessed on June 14, 2022.
[5] Mayo Clinic. Chemotherapy (Internet). Mayo Foundation for Medical Education and Research (MFMER), c2022. Available online: https://www.mayoclinic.org/tests-procedures/chemotherapy/about/pac-20385033#:~:text=Chemotherapy%20is%20a%20drug%20treatment,different%20chemotherapy%20drugs%20are%20available. Accessed on: June 14, 2022.
[6] Vighi G, Marcucci F, Sensi L, Di Cara G, Frati F. Allergy and the gastrointestinal system. Clin Exp Immunol. 2008;153 Suppl 1(Suppl 1):3-6. doi:10.1111/j.1365-2249.2008.03713.x
[7] Kadosh D and Geva-Zatorsky N. How do gut bacteria regulate our immune system? Front. Young Minds, 2021. 9:721325. doi: 10.3389/frym.2021.721325
[8] Martel, J., Chang, S.H., Ko, Y.F., Hwang, T.L., Young, J.D. and Ojcius, D.M., 2022. Gut barrier disruption and chronic disease. Trends in Endocrinology & Metabolism.
[9] Yoon MY, Yoon SS. Disruption of the Gut Ecosystem by Antibiotics. Yonsei Med J. 2018;59(1):4-12. doi:10.3349/ymj.2018.59.1.4
[10] Rinninella E, Raoul P, Cintoni M, Franceschi F, Miggiano GAD, Gasbarrini A, Mele MC. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms. 2019; 7(1):14. https://doi.org/10.3390/microorganisms7010014
[11] Zitvogel L, Galluzzi L, Viaud S, et al. Cancer and the gut microbiota: an unexpected link. Sci Transl Med. 2015;7(271):271ps1. doi:10.1126/scitranslmed.3010473
[12] Maier, L., Pruteanu, M., Kuhn, M. et al. Extensive impact of non-antibiotic drugs on human gut bacteria. Nature 555, 623–628 (2018). https://doi.org/10.1038/nature25979
[13] Villena J, Kitazawa H. Editorial: Immunobiotics-Interactions of Beneficial Microbes with the Immune System. Front Immunol. 2017;8:1580. Published 2017 Nov 17. doi:10.3389/fimmu.2017.01580
[14] Golkhalkhali B, Rajandram R, Paliany AS, Ho GF, Wan Ishak WZ, Johari CS, Chin KF. Strain-specific probiotic (microbial cell preparation) and omega-3 fatty acid in modulating quality of life and inflammatory markers in colorectal cancer patients: a randomized controlled trial. Asia Pac J Clin Oncol. 2018 Jun;14(3):179-191. doi: 10.1111/ajco.12758. Epub 2017 Aug 31. PMID: 28857425.
[15] Zaharuddin, L., Mokhtar, N.M., Muhammad Nawawi, K.N. et al. A randomized double-blind placebo-controlled trial of probiotics in post-surgical colorectal cancer. BMC Gastroenterol 19, 131 (2019). https://doi.org/10.1186/s12876-019-1047-4
[16] Zhang C., Björkman A., Cai K., Liu G., Wang C., Li Y., Xia H., Sun L., Kristiansen K., Wang J., et al. Impact of a 3-Months Vegetarian Diet on the Gut Microbiota and Immune Repertoire. Front. Immunol. 2018;9:908. doi: 10.3389/fimmu.2018.00908.
[17] Singh R.K., Chang H.-W., Yan D., Lee K.M., Ucmak D., Wong K., Abrouk M., Farahnik B., Nakamura M., Zhu T.H., et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017;15:73. doi: 10.1186/s12967-017-1175-y.
[18] Carbone C., Piro G., Di Noia V., D’Argento E., Vita E., Ferrara M.G., Pilotto S., Milella M., Cammarota G., Gasbarrini A., et al. Lung and gut microbiota as potential hidden driver of immunotherapy efficacy in lung cancer. Mediat. Inflamm. 2019;2019:1–10. doi: 10.1155/2019/7652014.
[19] Jin Y., Dong H., Xia L., Yang Y., Zhu Y., Shen Y., Zheng H., Yao C., Wang Y., Lu S. The Diversity of Gut Microbiome is Associated with Favorable Responses to Anti–Programmed Death 1 Immunotherapy in Chinese Patients with NSCLC. J. Thorac. Oncol. 2019;14:1378–1389. doi: 10.1016/j.jtho.2019.04.007.
[20] Wojas-Krawczyk K., Kalinka E., Grenda A., Krawczyk P., Milanowski J. Beyond PD-L1 Markers for Lung Cancer Immunotherapy. Int. J. Mol. Sci. 2019;20:1915. doi: 10.3390/ijms20081915.
[21] Villéger R., Lopès A., Carrier G., Veziant J., Billard E., Barnich N., Gagnière J., Vazeille E., Bonnet M. Intestinal Microbiota: A Novel Target to Improve Anti-Tumor Treatment? Int. J. Mol. Sci. 2019;20:4584. doi: 10.3390/ijms20184584.
[22] Gopalakrishnan V., Helmink B.A., Spencer C.N., Reuben A., Wargo J.A. The Influence of the Gut Microbiome on Cancer, Immunity, and Cancer Immunotherapy. Cancer Cell. 2018;33:570–580. doi: 10.1016/j.ccell.2018.03.015.
[23] Bertrand A., Kostine M., Barnetche T., Truchetet M.-E., Schaeverbeke T. Immune related adverse events associated with anti-CTLA-4 antibodies: Systematic review and meta-analysis. BMC Med. 2015;13:1–14. doi: 10.1186/s12916-015-0455-8.
[24] Elkrief A., Derosa L., Zitvogel L., Kroemer G., Routy B. The intimate relationship between gut microbiota and cancer immunotherapy. Gut Microbes. 2019;10:424–428. doi: 10.1080/19490976.2018.1527167.
[25] Soldati L., Di Renzo L., Jirillo E., Ascierto P.A., Marincola F.M., De Lorenzo A. The influence of diet on anti-cancer immune responsiveness. J. Transl. Med. 2018;16:1–18. doi: 10.1186/s12967-018-1448-0.
[26] Marinelli L., Tenore G.C., Novellino E. Probiotic species in the modulation of the anticancer immune response. Semin. Cancer Biol. 2017;46:182–190. doi: 10.1016/j.semcancer.2017.08.007.
[27] Dubin K., Callahan M.K., Ren B., Khanin R., Viale A., Ling L., No D., Gobourne A., Littmann E., Huttenhower B.R.C., et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpoint-blockade-induced colitis. Nat. Commun. 2016;7:10391. doi: 10.1038/ncomms10391.
[28] Li W., Deng Y., Chu Q., Zhang P. Gut microbiome and cancer immunotherapy. Cancer Lett. 2019;447:41–47. doi: 10.1016/j.canlet.2019.01.015.
[29] Vétizou M., Daillère R., Zitvogel L. Rôle du microbiote intestinal dans la réponse aux thérapies anti-tumorales. Biol. Aujourd’hui. 2017;211:51–67. doi: 10.1051/jbio/2017009.
[30] Bhatt A.P., Redinbo M.R., Bultman S.J. The role of the microbiome in cancer development and therapy. CA Cancer J. Clin. 2017;67:326–344. doi: 10.3322/caac.21398.
[31] Mohamad Nor MH, Ayob N, Mokhtar NM, Raja Ali RA, Tan GC, Wong Z, Shafiee NH, Wong YP, Mustangin M, Nawawi KNM. The Effect of Probiotics (MCP® BCMC® Strains) on Hepatic Steatosis, Small Intestinal Mucosal Immune Function, and Intestinal Barrier in Patients with Non-Alcoholic Fatty Liver Disease. Nutrients. 2021; 13(9):3192. https://doi.org/10.3390/nu13093192
[32] Baruch EN et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science, 2020. 371: 602-609. https://doi.org/10.1126/science.abb5920
[33] Tan CK, Said S, Rajandram R, Wang Z, Roslani AC, Chin KF. Pre-surgical Administration of Microbial Cell Preparation in Colorectal Cancer Patients: A Randomized Controlled Trial. World J Surg. 2016 Aug;40(8):1985-92. doi: 10.1007/s00268-016-3499-9. PMID: 27098538.
[34] Firouzi S, Mohd-Yusof BN, Majid HA, Ismail A, Kamaruddin NA. Effect of microbial cell preparation on renal profile and liver function among type 2 diabetics: a randomized controlled trial. BMC Complement Altern Med. 2015 Dec 12;15:433. doi: 10.1186/s12906-015-0952-5. PMID: 26654906; PMCID: PMC4676823.
Your email address cannot be published. Required fields are marked*
Comments:0
No comments